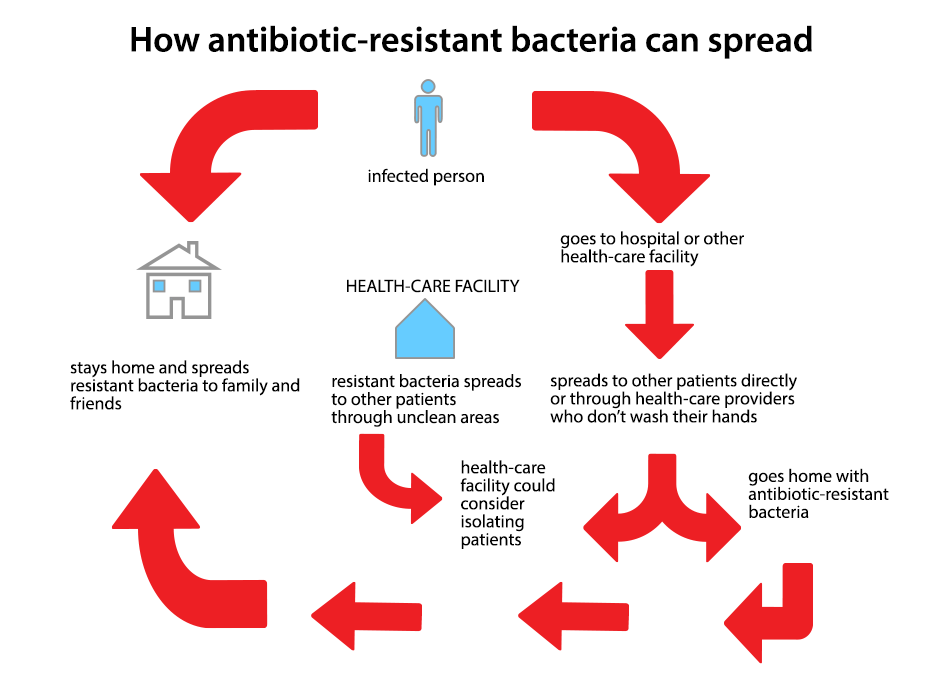
Virginia Mason fails to report "superbug" outbreak
/The Seattle outbreak entailed dangerous or deadly infections spread by tainted dueodenoscopes, even after cleaning according to Manufacturer’s guidelines. This latest report from the Washington State Department of Health faulted Virginia Mason for failing to properly notify state health officials of the outbreak, via a required report that tracks such dangerous events – Virginia Mason detected 39 infections and 18 deaths linked to the outbreak. Virginia Mason issued a statement explaining how the reporting of the incident came to be. But for the readers of our Products Blog, it's important to note that Virginia Mason was not found to be at fault for its handling of the procedures conducted here, including how it cleaned the duodenoscopes in accordance with the manufacturer’s guidelines. This is to say that the report confirms that this is first and foremost a product design (and perhaps product warning) problem. Virginia Mason may well be liable in other ways for its handling of the case, but that will play out in court and indeed Virginia Mason is facing lawsuits by families of patients harmed by the bacteria. Virginia Mason has in turn sued the manufacturer of the devices. The fact that patients could contract a nightmare bacteria this way is an outrage; we can only hope that the lawsuits will help to ensure that dangerous products are not brought into our hospitals, and will educate our hospitals on how to handle such situations should they arise again.